
Moreover, companies need to perform processes and procedures proactively to find comprehensive issues. The immediate correction and containment required problems should be completed rapidly to avoid further disruption. Under this phase, the risks should be verified and validated. It provides end-to-end traceability of the closed-loop quality processes which allows to track each and every root cause of the problems. The root cause determination process is encouraged by the quality management systems. 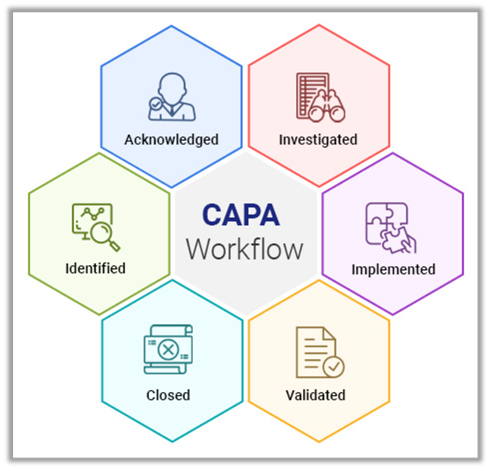
The next step of the CAPA quality management system is performing rapid investigations and root cause analysis of these problems with several methods. Investigation and Root Cause Determination: And the problems are separated as a low- risk issues and high-risk issues which need urgency in action. 
The problems identified from the risk analysis should be mentioned with the CAPA timeline. By using these details, a risks analysis should be performed based on the compliance risk. And it should be documented as the CAPA detection phase with complete details including who, what, when, where, and how. Initially, the problem should be identified by the organizations. 5 Fundamental working principles of CAPA Quality Management System 1. QMS with C APA management system is developed with an intimate understanding of five significant principles of the CAPA quality process. A CAPA quality system is considered to be a part of ISO compliance and plays a vital role in the TQM practices of companies that want to achieve goals with zero defects.ĬAPA integrated Quality management software is built to support companies fulfill the FDA guidelines by simplifying their Corrective and preventive action processes. Corrective actions and preventive actions help to address, rectify and eliminate all your unpredictable issues from ever happening again. CAPA:ĬAPA quality system is the most essential element for a complete quality management program. To accomplish or sustain these success factors every company requires a proper quality management system. Every regulated company concentrates on effective quality systems in order to maintain and overhead competition in the market, customer satisfaction, and cost control.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
